Self-Assembled Nanomaterials for Chronic Skin Wound Healing

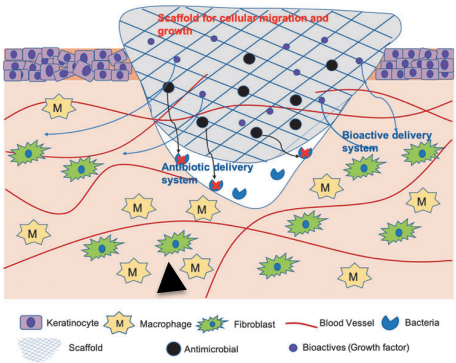
Chronic wounds can be caused by several underlying molecular and cellular pathophysiological mechanisms, including poor vascularization, excessive extracellular matrix (ECM) degradation by proteases, decreased growth factor activity, and bacterial infection. More effective wound therapies need to address one or more of these mechanisms to significantly advance wound care.
Self-assembled nanomaterials may provide new therapeutic options for chronic wound healing applications as those materials generally exhibit excellent biocompatibility and can bear multiple functionalities, such as ECM-mimicking properties, drug delivery capabilities, and tunable mechanics. Furthermore, self-assembled nanomaterials can be produced at low cost, and owing to their ability to self-organize, generate complex multifunctional structures that can be tailored to the varying sizes and shapes of chronic wounds. Self-assembled nanomaterials have been engineered to serve as wound dressings, growth factor delivery systems, and antimicrobials.
As there are many different types of self-assembled nanomaterials, which in turn have different mechanisms of self-assembly and physiochemical properties, one type of self-assembled nanomaterials may not be sufficient to address all underlying mechanisms of chronic wounds. However, self-assembled nanomaterials can be easily tailored, and developing multifunctional self-assembled nanomaterials that can address various targets in chronic wounds will be needed.
Future studies should investigate combinations of various self-assembled nanomaterials to take full advantage of their multifunctional properties.
Read the full article to know more.
Share this with your circle